Cydonia Quest The Occasional Journal C Entry No. 5 - 1st March 2004 Weird Science Invades Mars - Part Two In Steven Squyre's interview with Linda Moulton Howe he not only seemed hostile to the possibility of life on Mars but also to the presence of liquid water. Professor Squyres made great play of the fact that liquid water had not yet been found conclusively on the Martian surface. However, there is a growing suspicion that the "brine mud" that has been observed at the Spirit and Opportunity landing sites is often in a damp rather than a frozen state. This page of the Occasional Journal will return to this issue later. For the moment I want to deal with the weird "science" that is still heard on television from science commentators that the air pressure on Mars is too low to allow water to exist on or near the surface in its liquid state. As is well known the lower the pressure water is under the lower the temperature at which it boils away into water vapour. Once the pressure falls to six millibars or less the boiling point of water coincides with the temperature at which ice melts. So if we attempted to melt water ice that was under a pressure of less than six millibars it would boil straight from its frozen state into water vapour without ever having been liquid. Now it just happens that the average air pressure on the Martian surface is six millibars. So on the face of it liquid water seems to be an impossibility. However, as on Earth, Martian air pressure varies with altitude. The air pressure at the lowest points on Mars can be over 12 millibars. In fact air pressures of above 6 millibars were recorded at the two Viking lander (1976) and Pathfinder (1997) landing sites. At the locations on Mars where air pressure is greatest pure liquid water can exist on the surface at temperatures of between 0 oC and 10 oC without boiling away. Landers on Mars have measured maximum ground and soil temperatures higher than 20 0C, so there are no obstacles to ice melting.
|
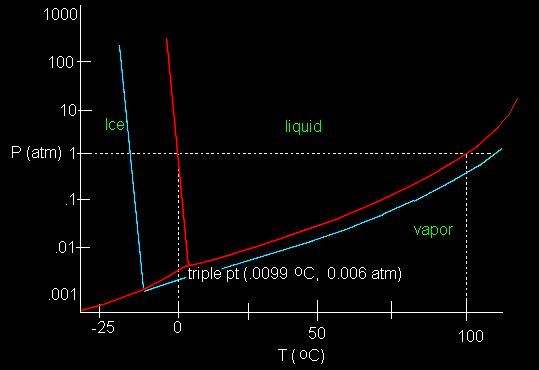
Of course it is well known that water in which impurities have been dissolved both freezes at a lower temperature and boils at a higher temperature at any given air pressure. The Viking landers measured concentrations of salts of between 10% and 20% of the soil volume at their landing sites. So there is plenty of scope for ground water to turn to salty brine - especially as there are no rains on Mars to dilute salty water as on Earth. C ccccccccccccc c The previous chart shows what happens to water when it is subject to different combinations of air pressure and temperature. The divisions shown in red show how pure water can be changed from ice, to liquid, to vapour at pressures above 6 millibars (0.006 Earth atmospheric pressure) by simply varying the temperature. At pressures below 6 millibars it shows that water can only exist as ice or vapour. (The triple point on the chart where the three divisions meet is a curious zone where water can either be ice, liquid or vapour as the "mood" takes it). The blue lines on the chart show the effects of a salty solution of unknown concentration. It can be seen that at the Martian average air pressure of 6 millibars there is suddenly a whole range of temperatures above and below zero degrees centigrade at which this salty water can be liquid, because the freezing point has been lowered and the boiling point increased. In fact salty water at the lowest altitudes on Mars would not even boil away at the maximum ground temperatures and might never freeze during the day at locations in the tropics. We know from the sensors of the Mars Odyssey that there is a lot of hidden water well away from the poles - so physics says there is a lot of liquid water hiding on Mars if it is salty. For a more detailed discussion about how salts would affect the existence of liquid water on Mars the reader is recommended to read this excellent NASA article - just click the following "stargate" µµµ. "The way the surface has responded is bizarre," said lead rover scientist Steve Squyres of Cornell University, at the Jet Propulsion Laboratory (JPL), which is managing the mission. "I don't understand it. I don't know anybody on my team who understands it. . . . It looks like mud, but it can't be mud." c cccccccc c One thing that has startled members of the Mars rovers team has been the stickiness of the soil at the Spirit and Opportunity landing sites. In fact as the quote from the team's Principle Investigator, Steven Squyres, makes clear - squeeze it and it acts just like damp earth. In a matter of days the team members were putting forward the theory that the soil was sticky because of salt left behind by brine that was evaporating up through the soil by capillary action. Vapour does not carry salt, so the liquid brine must be getting to the surface before it is evaporates and leaves behind this sticky salt. The image above has been processed by Keith Laney using raw image data from the Spirit rover. It was while I was perusing this image that I noticed that the retraction of the lander airbags had pulled a rock out of the ground in such a way as to expose its underside. Rather than being dulled by the adhesion of the sticky soil, the underside of this rock gleamed with a wet look as shown in the enlargement below. (More of Keith Laney's processed colour images from Spirit and Opportunity can be found at his website here µµµ ). C cccccccccccccccc
C Conspiracy theorists may be interested to know that this turns out to be exactly the same rock that the Enterprise Mission believes was deliberately blurred in a JPL processed version of the image in which this damp looking sheen is missing, (see µµµ). With the increasing evidence that the soils at the two landing sites may be slightly damp from salty water it is hardly surprising that speculation is growing again about the possibility of Martian life. Gilbert Levin, the designer of the life tests on the Viking landers, has always maintained that the Viking missions did find life and that the degree to which geologist dominated JPL discredited the results was unreasonable. In such a "political" atmosphere, the discovery of liquid water and the increased scope for the existence of life might be highly embarrassing to the JPL old guard. (Readers are recommended to read the article at the other end of this "stargate" µµµ). We will just have to see how this salty tale develops..... c µ Click this "Stargate" to return to Journal main page µ Return to the main Mars page
|